![EtO)3Si(CH2)3NH3+][CH3COO−] as a novel basic ionic liquid catalyzed green synthesis of new 2-(phenylsulfonyl)-1H-benzo[a]pyrano[2,3-c]phenazin-3-amine derivatives - ScienceDirect EtO)3Si(CH2)3NH3+][CH3COO−] as a novel basic ionic liquid catalyzed green synthesis of new 2-(phenylsulfonyl)-1H-benzo[a]pyrano[2,3-c]phenazin-3-amine derivatives - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022286022002319-gr11.jpg)
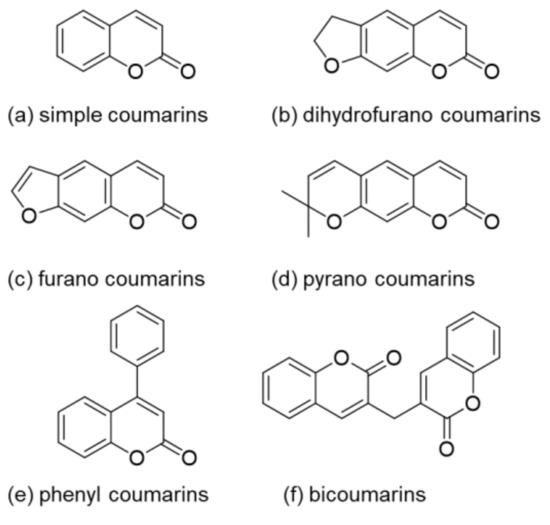
EtO)3Si(CH2)3NH3+][CH3COO−] as a novel basic ionic liquid catalyzed green synthesis of new 2-(phenylsulfonyl)-1H-benzo[a]pyrano[2,3-c]phenazin-3-amine derivatives - ScienceDirect
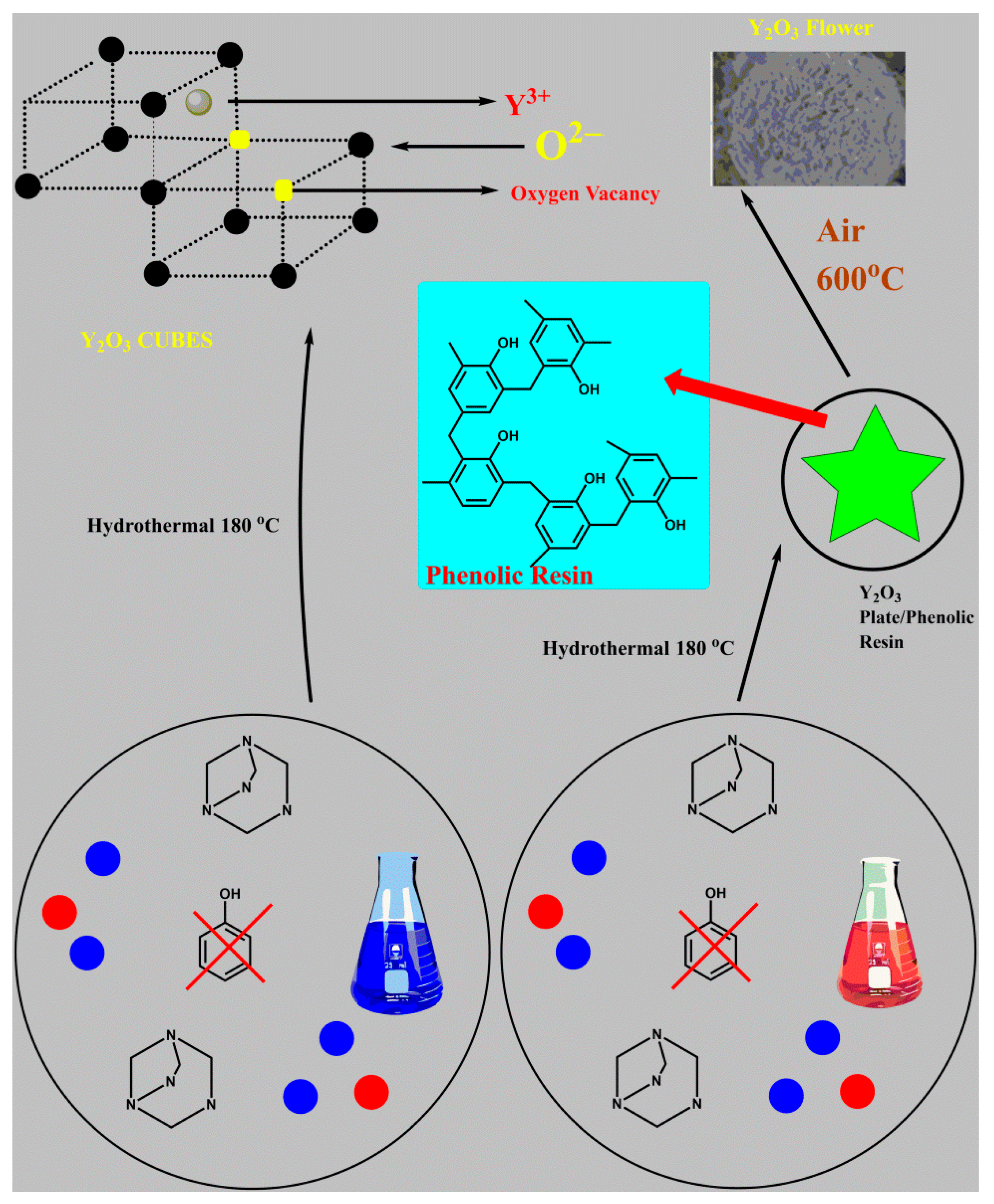
Applied Sciences | Free Full-Text | Yttrium Oxide Nanoparticle Synthesis: An Overview of Methods of Preparation and Biomedical Applications
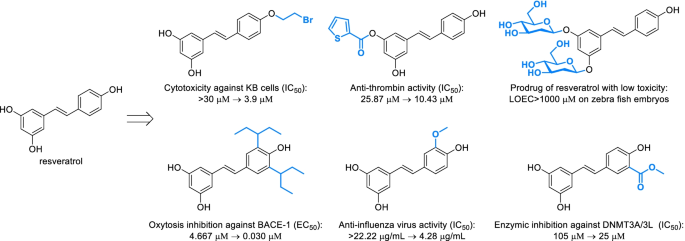
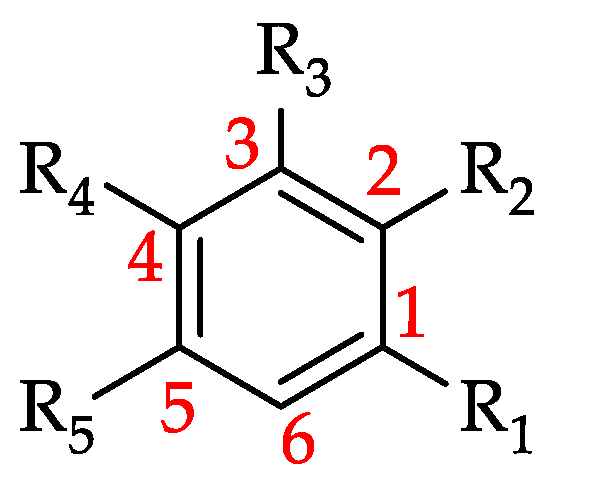
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink

Biosynthesis pathway of phenolic acids and stilbene from phenylalanine. | Download Scientific Diagram

Nickel(II)‐Catalyzed Diastereo‐ and Enantioselective Michael/ Hemiacetalization Cascade Reaction of α‐Ketoesters with 2‐(2‐Nitrovinyl) phenols - Chen - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library
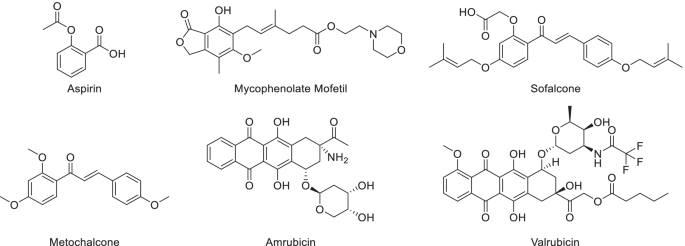
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink
Foods | Free Full-Text | A Comprehensive Survey of Phenolic Constituents Reported in Monofloral Honeys around the Globe

Bis(amino)cyclopropenium Ion as a Hydrogen-Bond Donor Catalyst for 1,6-Conjugate Addition Reactions | The Journal of Organic Chemistry
![Substituted 1,1'-biphenyl Compounds, Analogues Thereof, And Methods Using Same Bi; Yingzhi ; et al. [Arbutus Biopharma Corporation] Substituted 1,1'-biphenyl Compounds, Analogues Thereof, And Methods Using Same Bi; Yingzhi ; et al. [Arbutus Biopharma Corporation]](https://uspto.report/patent/app/20210052585/US20210052585A1-20210225-C00002.png)
Substituted 1,1'-biphenyl Compounds, Analogues Thereof, And Methods Using Same Bi; Yingzhi ; et al. [Arbutus Biopharma Corporation]
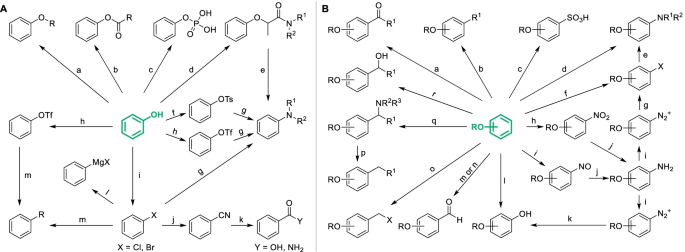
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink
![Practical Asymmetric Synthesis of (S)-4-Ethyl-7,8-dihydro-4-hydroxy-1H- pyrano[3,4-f]indolizine- 3,6,10(4H)-trione, a Key Intermediate for the Synthesis of Irinotecan and Other Camptothecin Analogs | The Journal of Organic Chemistry Practical Asymmetric Synthesis of (S)-4-Ethyl-7,8-dihydro-4-hydroxy-1H- pyrano[3,4-f]indolizine- 3,6,10(4H)-trione, a Key Intermediate for the Synthesis of Irinotecan and Other Camptothecin Analogs | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo970173f/asset/images/large/jo970173fn00001.jpeg)
Practical Asymmetric Synthesis of (S)-4-Ethyl-7,8-dihydro-4-hydroxy-1H- pyrano[3,4-f]indolizine- 3,6,10(4H)-trione, a Key Intermediate for the Synthesis of Irinotecan and Other Camptothecin Analogs | The Journal of Organic Chemistry
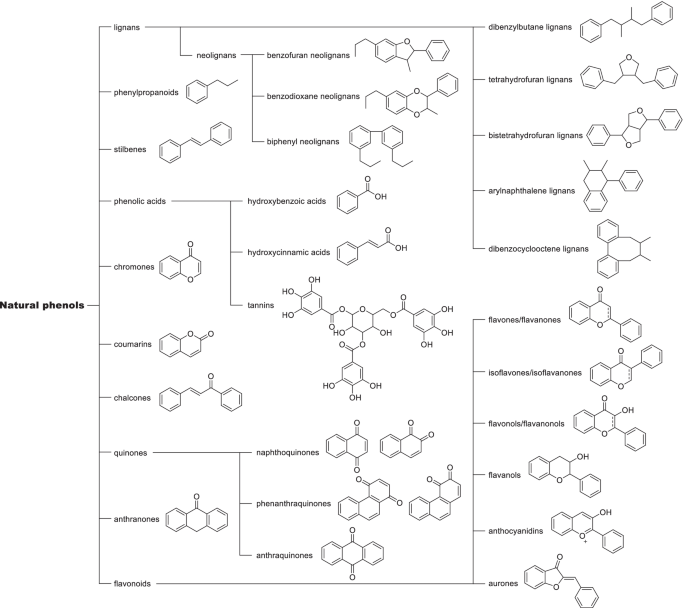
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink

Synthesis of Indolequinones from Bromoquinones and Enamines Mediated by Cu(OAc)2·H2O | The Journal of Organic Chemistry

Rhodium(I)-Catalyzed Cyclization Reaction of o-Alkynyl Phenols and Anilines. Domino Approach to 2,3-Disubstituted Benzofurans and Indoles | Organic Letters

Efficient Generation of ortho-Quinone Methide: Application to the Biomimetic Syntheses of (±)-Schefflone and Tocopherol Trimers | Organic Letters

Investigation on the reactions of o‐hydroxybenzyl alcohols with vinyl ethers under acidic and/or thermal conditions - Kumbaraci - 2009 - Journal of Heterocyclic Chemistry - Wiley Online Library

Trisubstituted Aziridine Ring-Opening by Phenol Derivatives: Stereo- and Regioselective Formation of Chiral Tertiary Alkyl-Aryl Ethers | Organic Letters
![Intramolecular Diels–Alder Cycloaddition of Furan-Derived β-Enamino Diketones: An Entry to Diastereoselective Synthesis of Polycyclic Pyrano[3,2-c]quinolin-5-one Derivatives | The Journal of Organic Chemistry Intramolecular Diels–Alder Cycloaddition of Furan-Derived β-Enamino Diketones: An Entry to Diastereoselective Synthesis of Polycyclic Pyrano[3,2-c]quinolin-5-one Derivatives | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.1c03163/asset/images/medium/jo1c03163_0010.gif)
Intramolecular Diels–Alder Cycloaddition of Furan-Derived β-Enamino Diketones: An Entry to Diastereoselective Synthesis of Polycyclic Pyrano[3,2-c]quinolin-5-one Derivatives | The Journal of Organic Chemistry