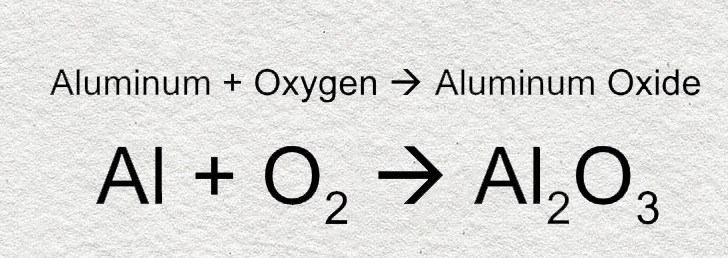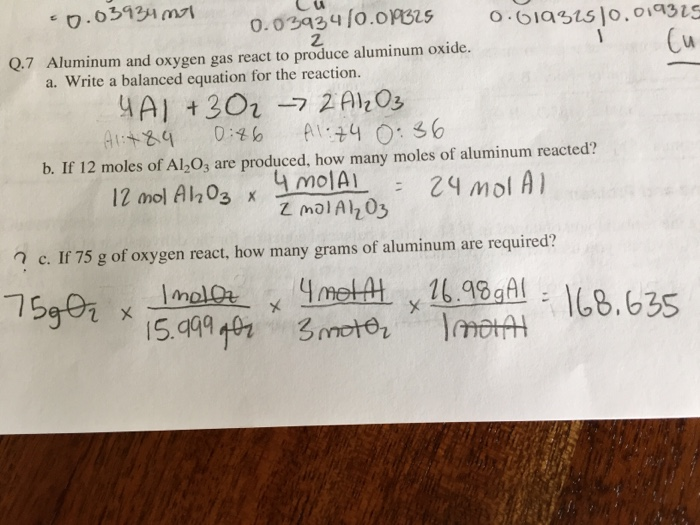SOLVED: QUESTiON 22 Aluminum reacts with oxygen to give aluminum oxide: Al(s) 302(g) Al203(s) If you have 6.0 moles of Al, you need 3.0 moles of 02 for complete reaction and produce

Practice Write a balanced formula equation for: aluminum + oxygen aluminum oxide Al +Al OO2 →O2 →Al 2 O ppt download

SOLVED:Aluminum reacts with oxygen to give aluminum oxide. 4 Al(s)+3 O2(g) ⟶2 Al2 O3(s) What amount of O2, in moles, is needed for complete reaction with 6.0 mol of Al? What mass
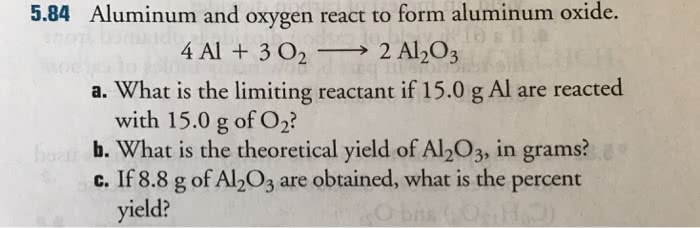
OneClass: Aluminum and oxygen react to form aluminum oxide. 4 Al + 3 O_2 rightarrow 2 Al_2O_3 a. What...

1 Review Aluminum reacts with oxygen to produce aluminum oxide Phosphoric acid, is produced through the reaction between tetraphosphorus decaoxide and. - ppt download

SOLVED: A 3.53-g sample of aluminum completely reacts with oxygen to form 6.67 g of aluminum oxide. Use this data to calculate the mass percent composition of aluminum in aluminum oxide.

SOLVED: Iow all work steps for full credit There are 10 problems - tolaling " to 20 point; points) Solid Aluminum reacts with oxygen gAs produce solid aluminum oxide Write - balanced