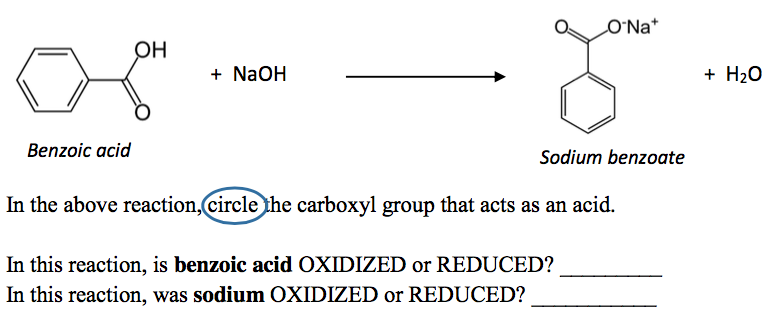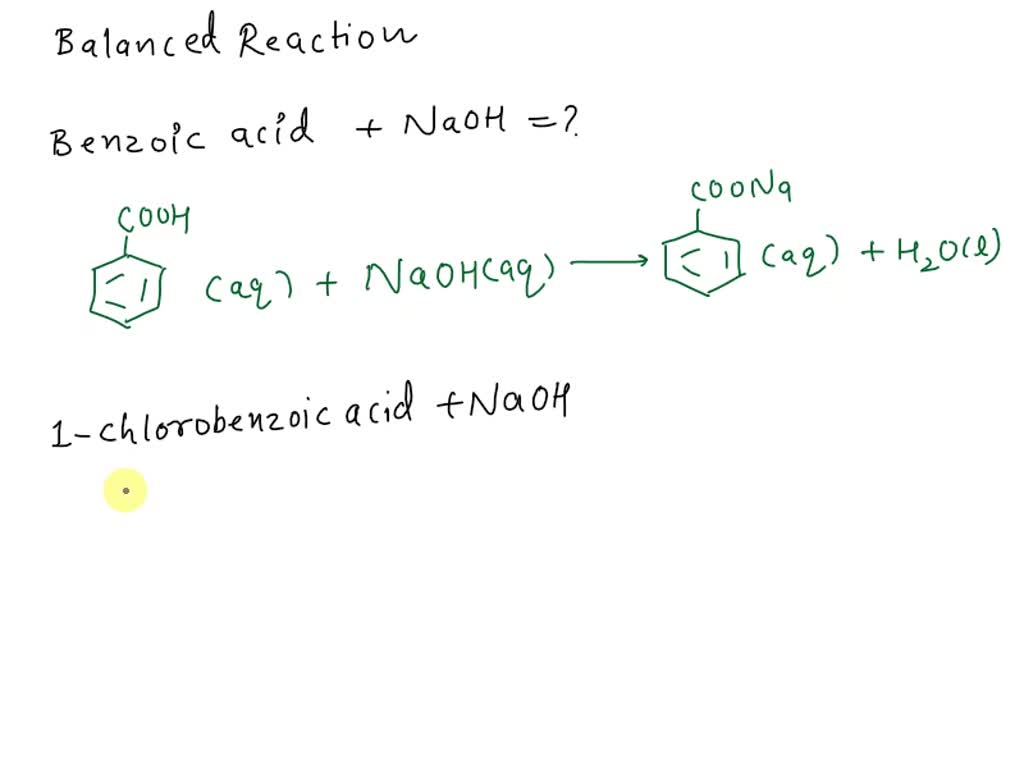![Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid](https://www.vedantu.com/question-sets/39d15196-f3b7-4d8c-bde9-5684a8a019909186147776397460082.png)
Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid
You are planning to purify an impure sample of benzoic acid, known to be contaminated with naphthalene. - Sarthaks eConnect | Largest Online Education Community

Synthesis of the internal standard 4-(methoxy-d 3 )-benzoic acid (2).... | Download Scientific Diagram
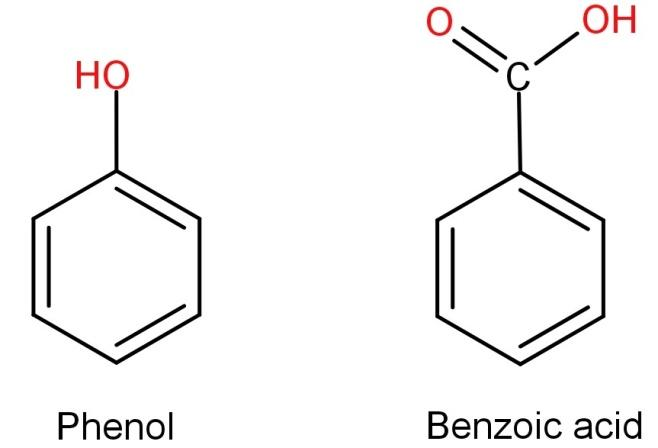
A mixture of phenol and benzoic acid will completely dissolve in an aqueous solution of?(A) HCl(B) NaCl(C) $NaHC{O_3}$ (D) NaOH

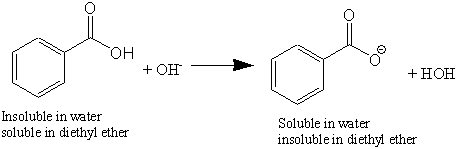
OneClass: Benzoic acid is soluble in diethyl ether but not water, however, benzoic acid is extracted ...

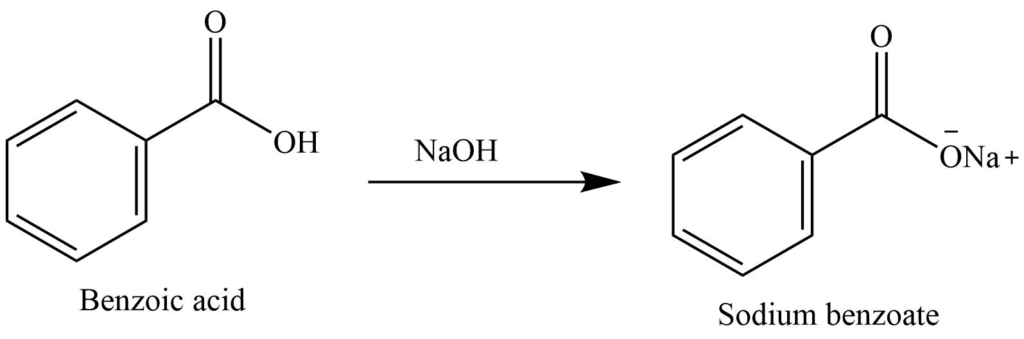
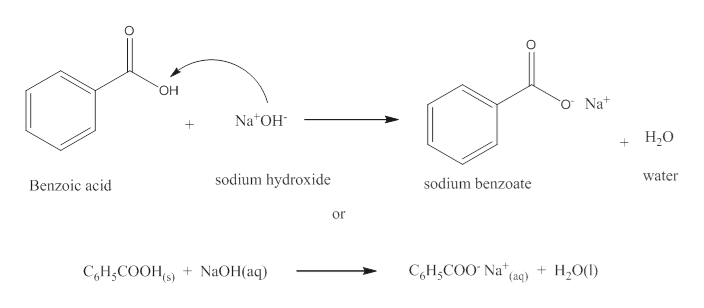
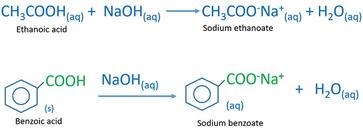
Write a balanced equation for the reaction of benzoic acid with hydroxide ion. Why is it necessary to extract the ether layer with sodium hydroxide? | Homework.Study.com

Draw the products of benzoic acid reacting with sodium hydroxide. Draw the products of the pyridine reacting with hydrochloric acid. Use the "+/-" button to add the charge (and H atom).