
List the characteristics of cork. How are they formed? Mention their role.a) Write the formula of (i) Magnesium hydroxide (ii) Hydrogen sulphide (iii) Potassium chloride (iv) Calcium oxide (v) Barium chloride (vi)
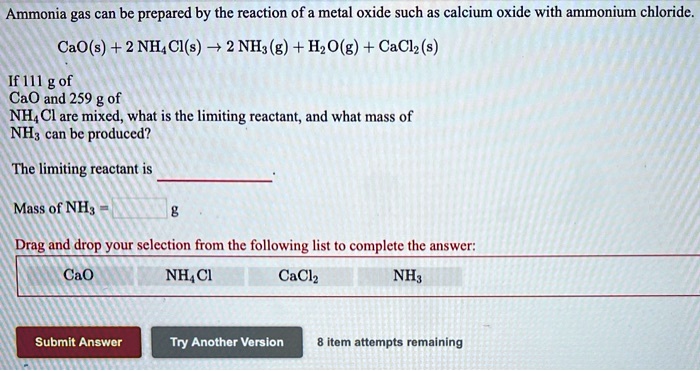
SOLVED: Ammonia gas can be prepared by the reaction of metal oxide such as calcium oxide with ammonium chloride. CaO(s) + 2 NH,CI(s) 2 NHs(g) + Hz(g) + CaClz (s) If I1
The remaining gas is mainly nitrogen containing some impurities of rare gases which cannot be removed by any chemical means as
![Why calcium ions do not form precipitate with aqueous ammonia [online video] – O Level Secondary Chemistry Tuition Why calcium ions do not form precipitate with aqueous ammonia [online video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2021/10/qa-aq-ammonia.jpg?w=986)
Why calcium ions do not form precipitate with aqueous ammonia [online video] – O Level Secondary Chemistry Tuition

Nitrogen has a triple bond which is very strong. :N:::N: Only at very high temperatures will it react with oxygen. This occurs in the combustion. - ppt download

SOLVED: Ammonia gas can be prepared by the reaction of a metal oxide such as calcium oxide with ammonium chloride: CaO(s) + 2 NH4 Cl(s) 72 NH (g) + HzO(g) + Ca

Toward the Mechanistic Understanding of the Additives' Role on Ammonium Nitrate Decomposition: Calcium Carbonate and Calcium Sulfate as Case Studies | ACS Omega

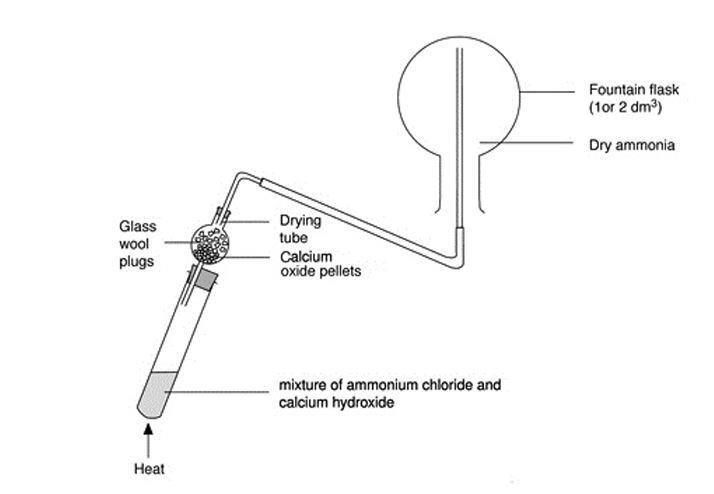
Calcium hydroxide and ammonium chloride react to give ammonia as per equation: Ca(OH)_(2) + 2 NH... - YouTube

Removal of Sulfate Ions from Calcium Oxide Precipitation Enrichment of a Rare Earth Leaching Liquor by Stirring Washing with Sodium Hydroxide | ACS Omega
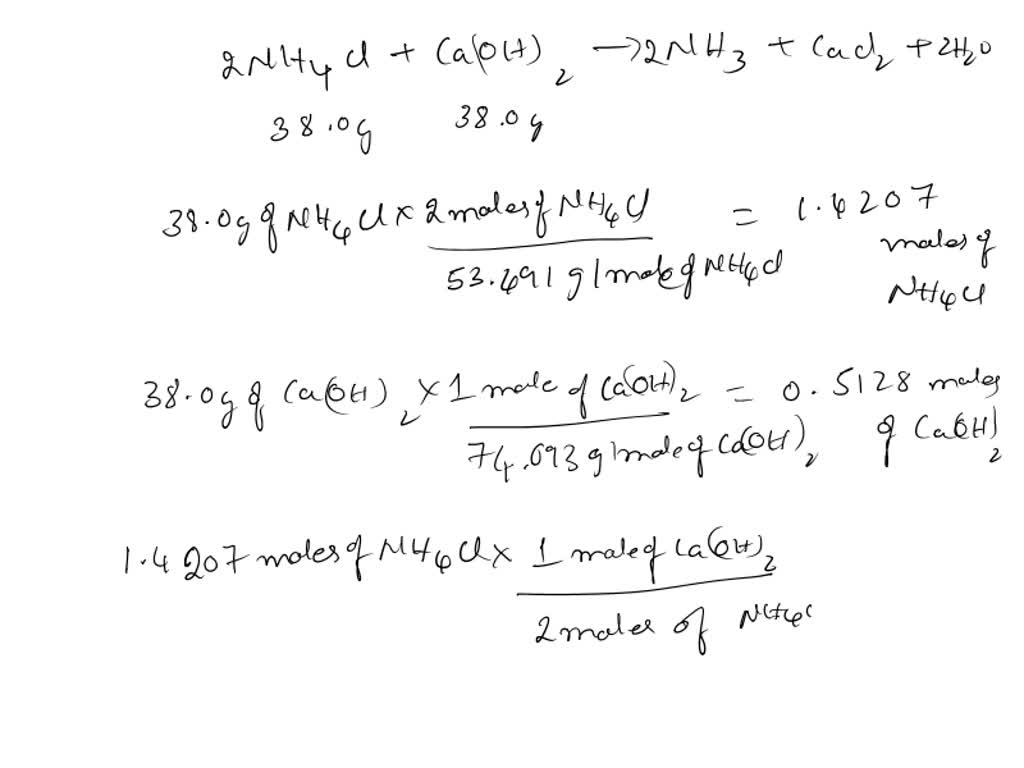
SOLVED: 2. Ammonia can be made by heating solid ammonium chloride with solid calcium oxide to form gaseous ammonia, solid calcium chloride, and water vapor.a. Write and balance the chemical equationb. How
![Why calcium ions do not form precipitate with aqueous ammonia [online video] – O Level Secondary Chemistry Tuition Why calcium ions do not form precipitate with aqueous ammonia [online video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2021/10/pic-4.jpg?w=1024)








.png)



