organic chemistry - Why can't alcohols form hydrogen-bonded dimers like carboxylic acids? - Chemistry Stack Exchange

Solid-State 17O NMR Study of Carboxylic Acid Dimers: Simultaneously Accessing Spectral Properties of Low- and High-Energy Tautomers | The Journal of Physical Chemistry A
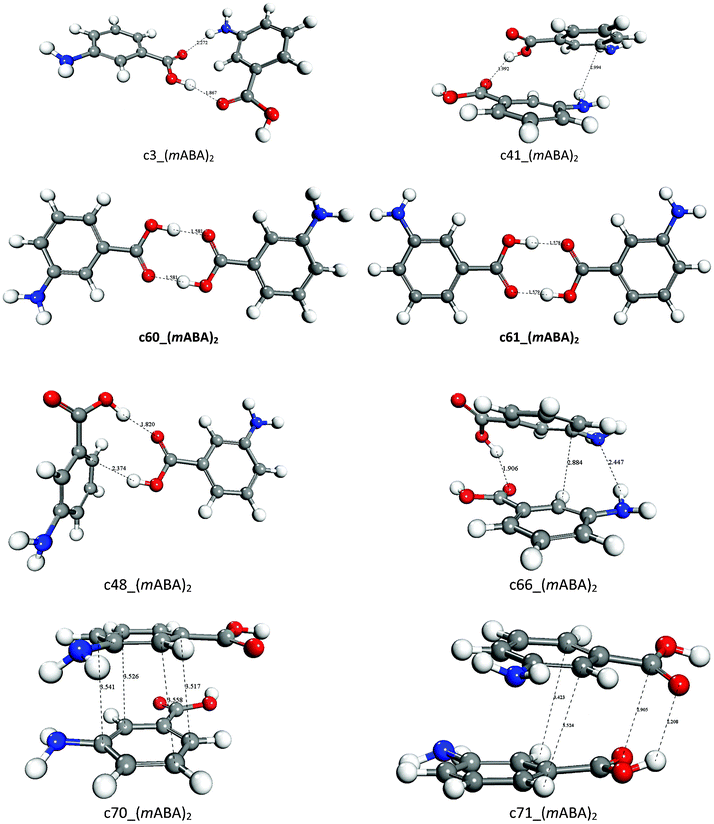
The molecular self-association of carboxylic acids in solution: testing the validity of the link hypothesis using a quantum mechanical continuum solva ... - CrystEngComm (RSC Publishing) DOI:10.1039/C3CE40539G

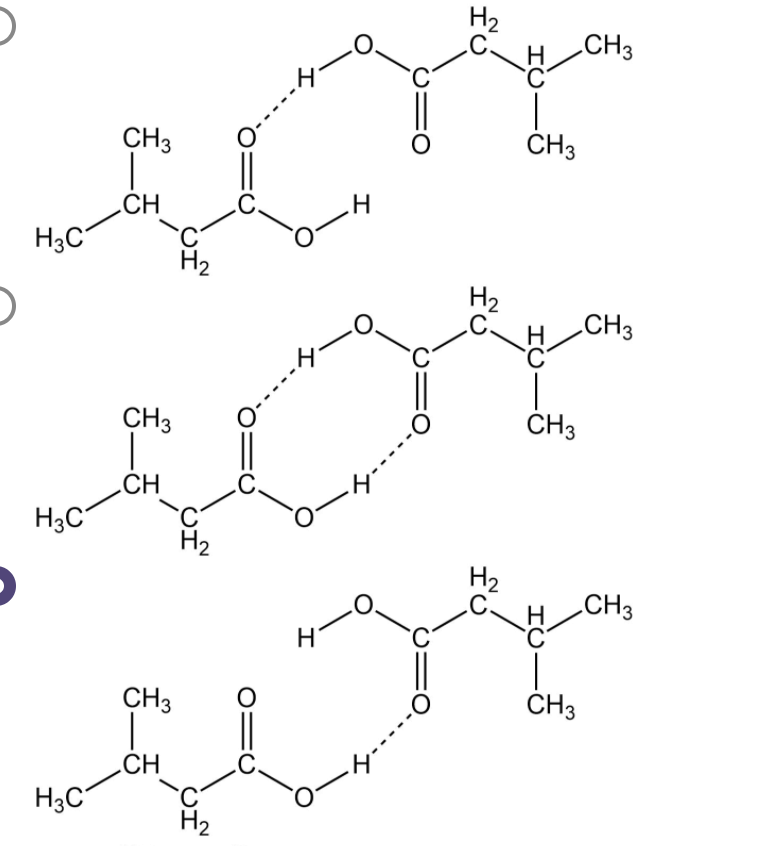
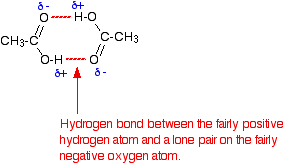
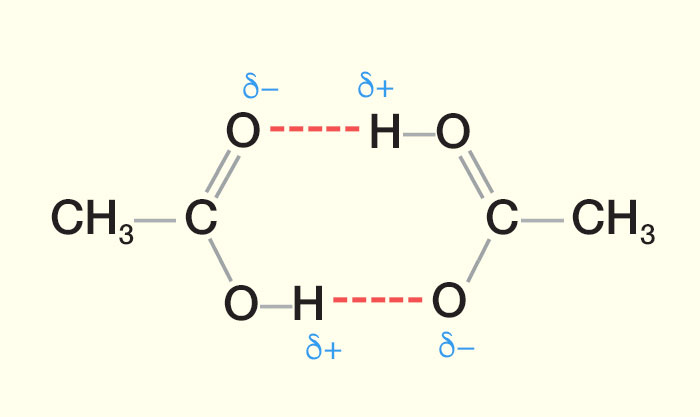
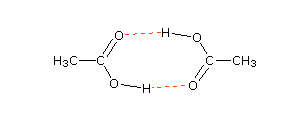
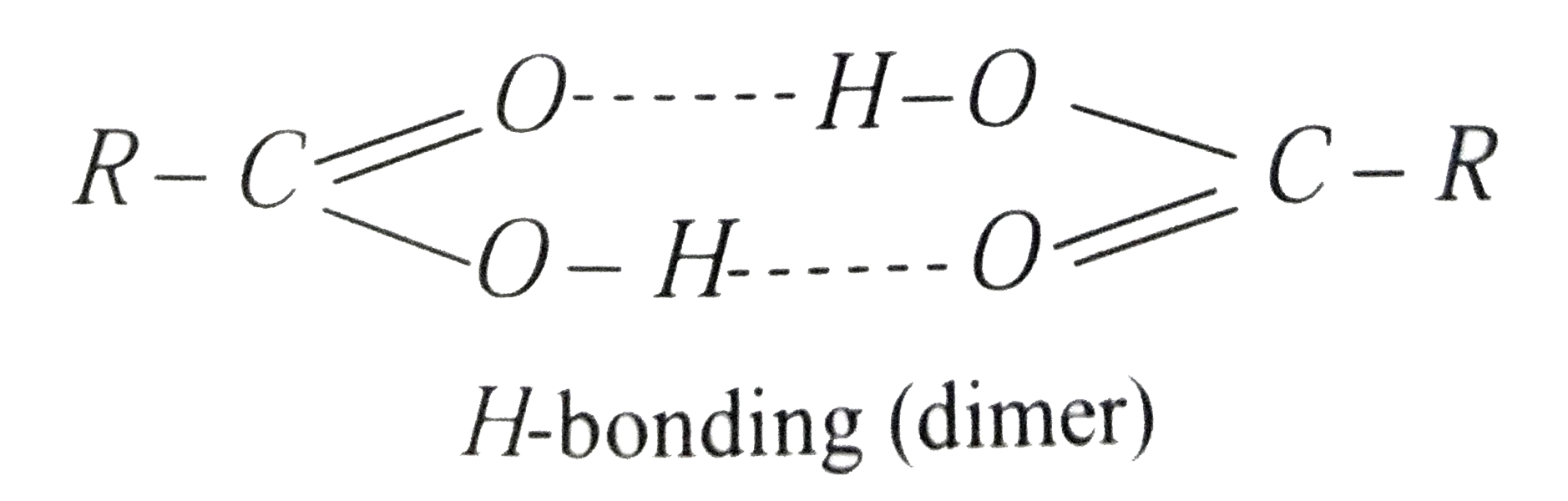
Carboxylic acids have higher than expected boiling points due to dimeric associations involving hydrogen bonding. The structure of 3-methylbutyric acid is shown below. Draw a second molecule of 3-meth | Homework.Study.com

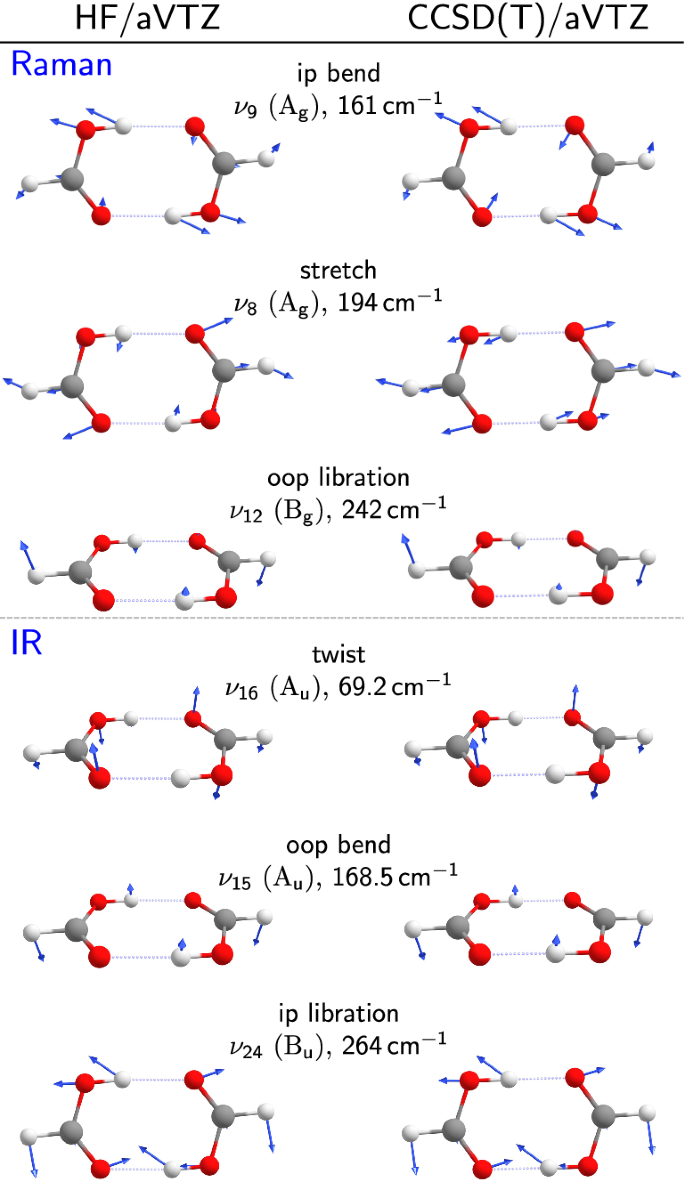
Vibrational exciton coupling in homo and hetero dimers of carboxylic acids studied by linear infrared and Raman jet spectroscopy: The Journal of Chemical Physics: Vol 149, No 10
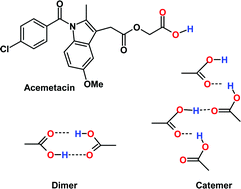
Acemetacin polymorphs: a rare case of carboxylic acid catemer and dimer synthons - CrystEngComm (RSC Publishing)

297175-66-7 | Amoxicilloic Acid Dimer (Mixture of Diastereomers) | (2S,4S)-2-((R)-1-((R)-2-Amino-2-(4-hydroxyphenyl)acetamido)-2-(((R)-2-((((2S,4S)-4-carboxy-5,5-dimethylthiazolidin-2-yl)methyl)amino)-1-(4-hydroxyphenyl)-2-oxoethyl)amino)-2-oxoethyl)-5 ...

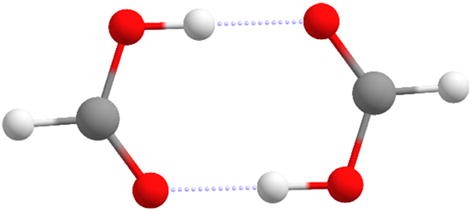
Encapsulated Carboxylic Acid Dimers with Compressed Hydrogen Bonds - Ajami - 2011 - Angewandte Chemie International Edition - Wiley Online Library

Carboxylic acid dimer O − H ··· O synthon of R 22 (8) ring motif and... | Download Scientific Diagram