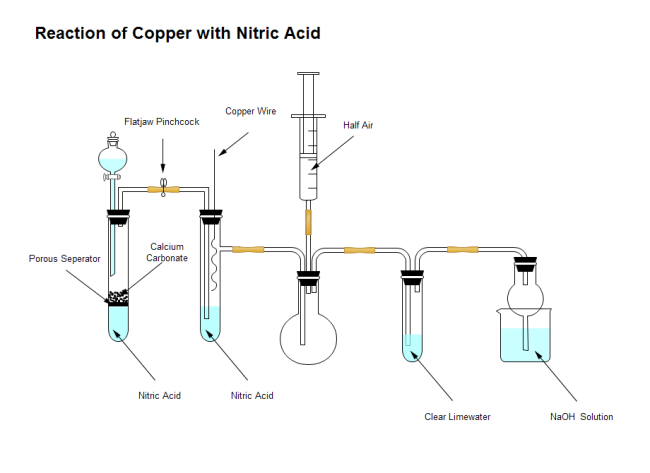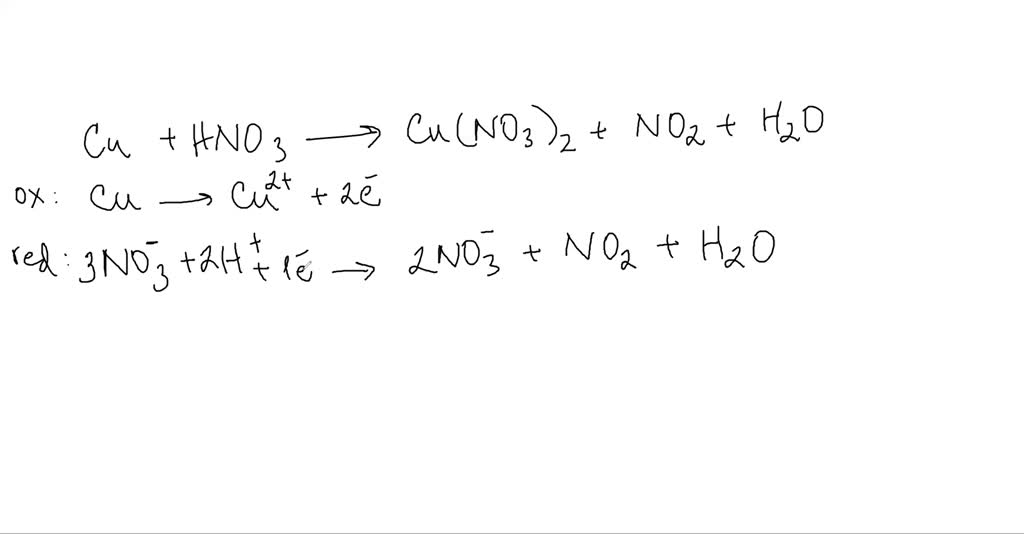
SOLVED:When solid copper is added to nitric acid, copper(II) nitrate, nitrogen dioxide, and water are produced. Write the balanced chemical equation for the reaction. List six mole ratios for the reaction.

تويتر \ Compound Interest على تويتر: "Reacting copper with concentrated nitric acid: http://t.co/CkQhEbTt5b"
Reaction of CuO with nitric acid, left to right: fuming, concentrated,... | Download Scientific Diagram

Reaction of copper with Nitric acid (HNO3), also known as aqua fortis and spirit of niter, is a highly corrosive mineral acid Stock Photo - Alamy

BALANCING EQUATIONS WHERE ELEMENTS APPEAR IN MORE THAN ONE PRODUCT SPECIES Example: Copper metal + Nitric Acid → Copper Nitrate + Nitrogen Dioxide + Water. - ppt video online download


2%20+%20NO%20+%20H2O%20reaction.jpg?ezimgfmt=rs:323x202/rscb1/ngcb1/notWebP)