Dehydroacetic acid and its sodium salt: Human health tier II assessment Chemicals in this assessment Preface

Integrated approaches to testing and assessment as a tool for the hazard assessment and risk characterization of cosmetic preservatives - Canavez - 2021 - Journal of Applied Toxicology - Wiley Online Library

A repeated dose 28-day oral toxicity study of sodium dehydroacetate (DHA-S) in Wistar rats - ScienceDirect
Dehydroacetic acid and its sodium salt: Human health tier II assessment Chemicals in this assessment Preface

A repeated dose 28-day oral toxicity study of sodium dehydroacetate (DHA-S) in Wistar rats - ScienceDirect

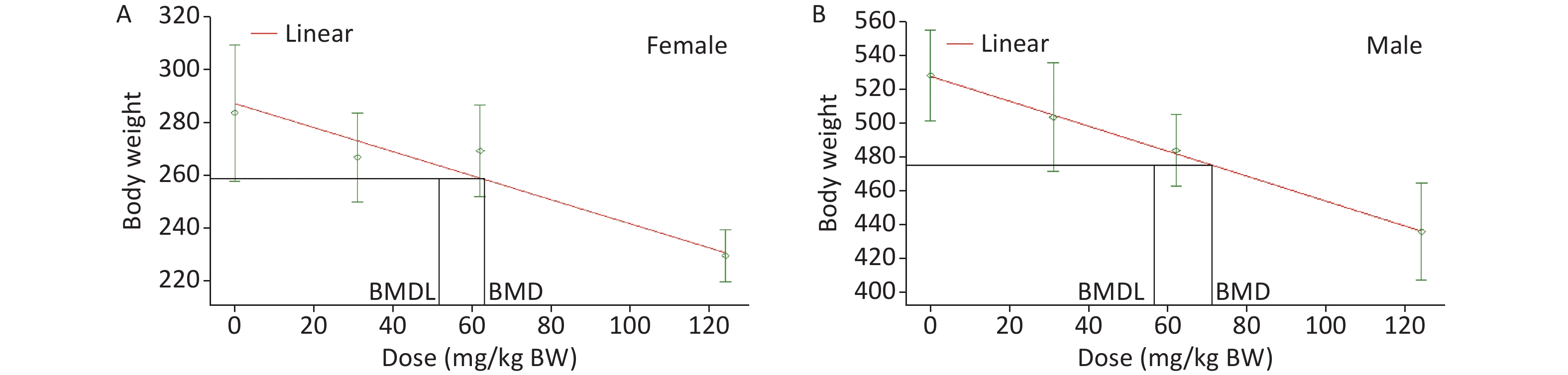
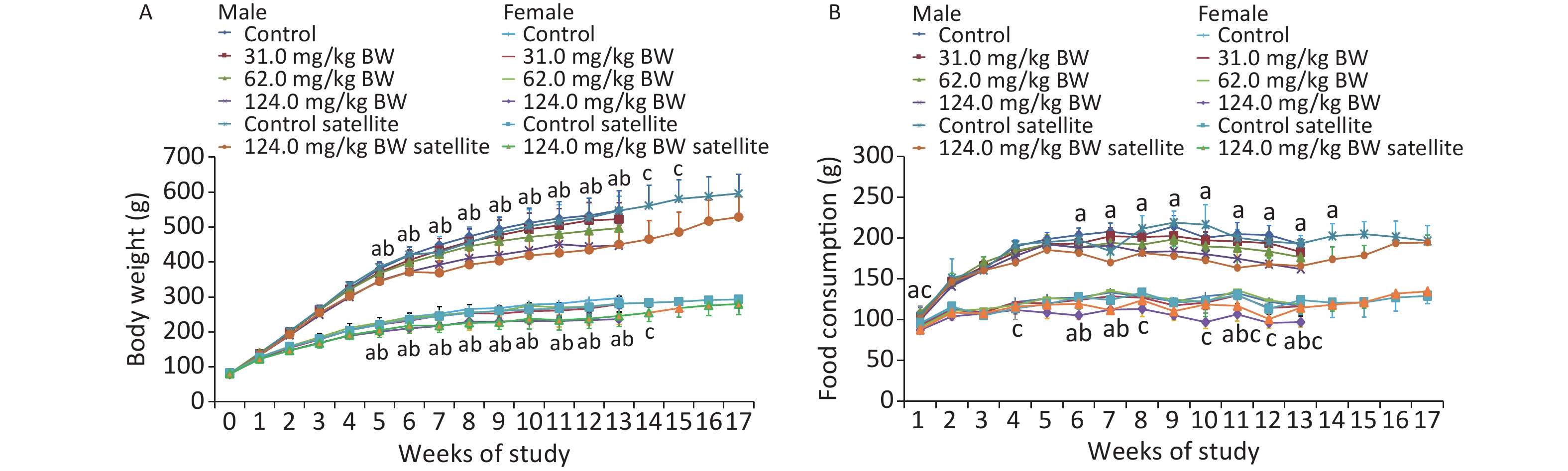
A repeated dose 28-day oral toxicity study of sodium dehydroacetate (DHA-S) in Wistar rats - ScienceDirect

A repeated dose 28-day oral toxicity study of sodium dehydroacetate (DHA-S) in Wistar rats - ScienceDirect
Dehydroacetic acid and its sodium salt: Human health tier II assessment Chemicals in this assessment Preface