![PDF] Experimental and Theoretical Studies of Oxalic Acid Dissociation in Water-Ethanol Solvents | Semantic Scholar PDF] Experimental and Theoretical Studies of Oxalic Acid Dissociation in Water-Ethanol Solvents | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/194587d529fcb2355d1a23e0723cca1d9c19adc3/4-Table3-1.png)
PDF] Experimental and Theoretical Studies of Oxalic Acid Dissociation in Water-Ethanol Solvents | Semantic Scholar
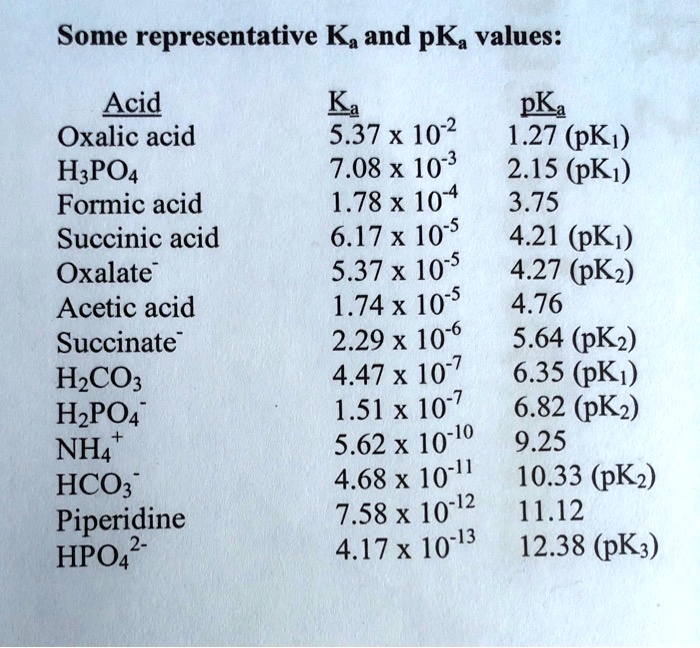
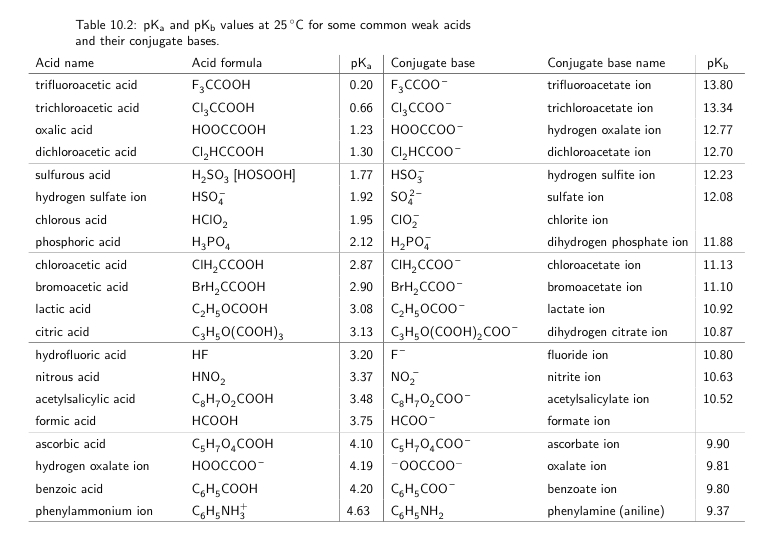
SOLVED: Some representative Ka and pKa values: Acid Oxalic acid HzPOa Formic acid Succinic acid Oxalate Acetic acid Succinate HzCOz HzPOA NH4' HCO; Piperidine HPOA2 Ka pKa 5.37 x 10-2 1.27 (pK,)

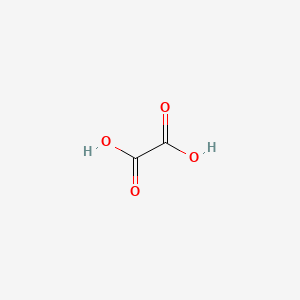
Three forms of oxalic acid in the aqueous solution with different pH value | Download Scientific Diagram

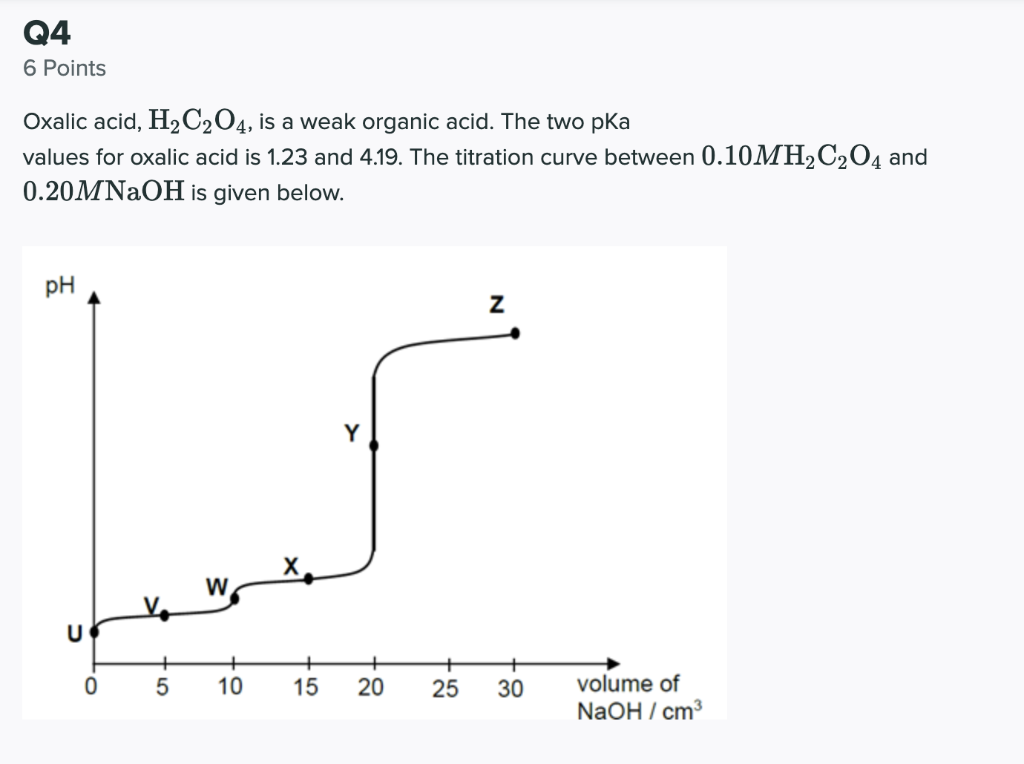
The correct order of pKa for the following acid:Oxalic acid pK1 Monalic acid pK2 Heptanedioic acid pK3 where pK1 , pK2 , pK3 are first ionization constants.
![SOLVED: What is the pH of a buffer composed of 0.10 M oxalic acid and 0.08 M oxalate ion? The pKa for oxalic acid is 1.23. pH = pKa log ([A-J[HA]) Use SOLVED: What is the pH of a buffer composed of 0.10 M oxalic acid and 0.08 M oxalate ion? The pKa for oxalic acid is 1.23. pH = pKa log ([A-J[HA]) Use](https://cdn.numerade.com/ask_images/08a0de4c43b94a8e8f10c82ca0bf2b68.jpg)
SOLVED: What is the pH of a buffer composed of 0.10 M oxalic acid and 0.08 M oxalate ion? The pKa for oxalic acid is 1.23. pH = pKa log ([A-J[HA]) Use

Development of Methods for the Determination of pKa Values - Jetse Reijenga, Arno van Hoof, Antonie van Loon, Bram Teunissen, 2013

Titrations and pKa CHEMISTRY Titrations and Pka predictions Titrations Method of quantitative/chemical analysis which can be used to determine. - ppt download

A Reliable and Efficient First Principles-Based Method for Predicting pKa Values. 2. Organic Acids | The Journal of Physical Chemistry A
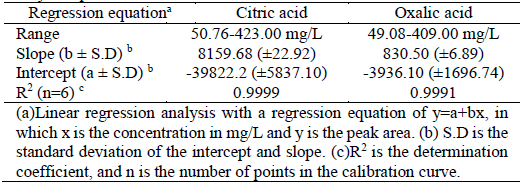
Determination of citric and oxalic acid in fungi fermentation broth through HPLC-DAD and solid-phase extraction
![PDF] Experimental and Theoretical Studies of Oxalic Acid Dissociation in Water-Ethanol Solvents | Semantic Scholar PDF] Experimental and Theoretical Studies of Oxalic Acid Dissociation in Water-Ethanol Solvents | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/194587d529fcb2355d1a23e0723cca1d9c19adc3/5-Table4-1.png)