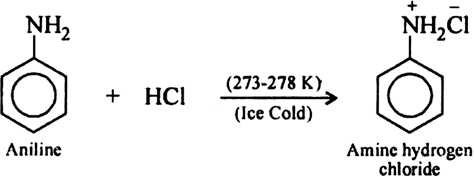
Write the reactions of (i) aromatic, (ii) aliphatic primary amines with nitrous acid. from Chemistry Amines Class 12 CBSE
Explain the action of nitrous acid with N – methyl aniline. - Sarthaks eConnect | Largest Online Education Community

The diazotization of aniline first involves the formation of NO+ (nitrosonium ion) by the dehydration of nitrous acid with sulfuric acid. The aniline nitrogen then acts as a nucleophile and eventually loses

Give balanced equations for the following reactions :(i) Ethylamine with nitrous acid.(ii) Diethyl ether with phosphorous pentachloride.(iii) Aniline with acetyl chloride.
What is the action of nitrous acid on aniline? - Sarthaks eConnect | Largest Online Education Community

The diazotization process. (a) The reaction of aniline (or other aryl... | Download Scientific Diagram
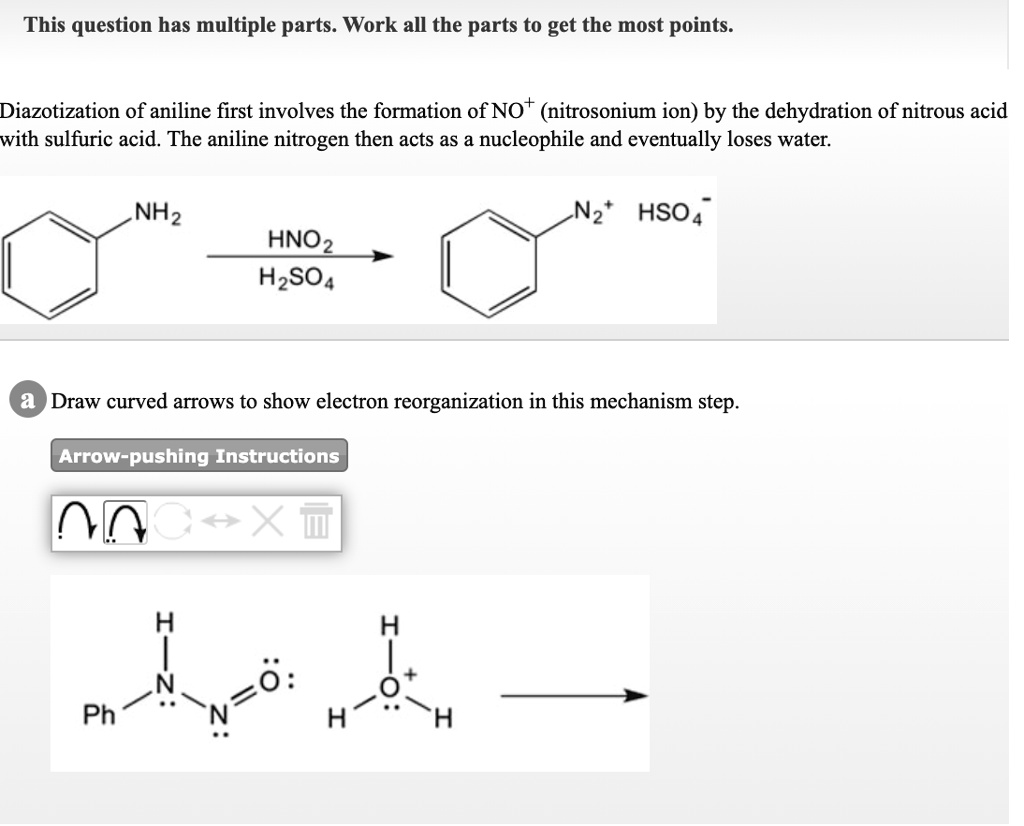
SOLVED: This question has multiple parts. Work all the parts to get the most points: Diazotization of aniline first involves the formation of NO+ (nitrosonium ion) by the dehydration of nitrous acid

organic chemistry - Which occurs first: nitration or oxidation of aniline by concentrated nitric acid? - Chemistry Stack Exchange

Give balanced equations for the following reactions:(i) Aniline is treated with nitrous acid and HCl at low temperature.(ii) Acetyl chloride is treated with ethyl alcohol.(iii) Formaldehyde is treated with ammonia.
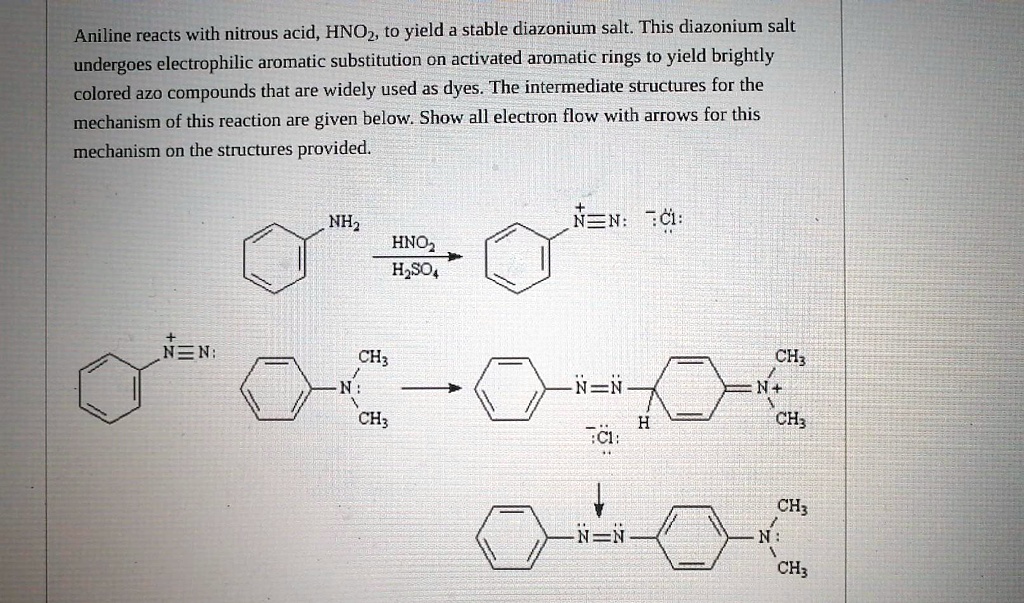
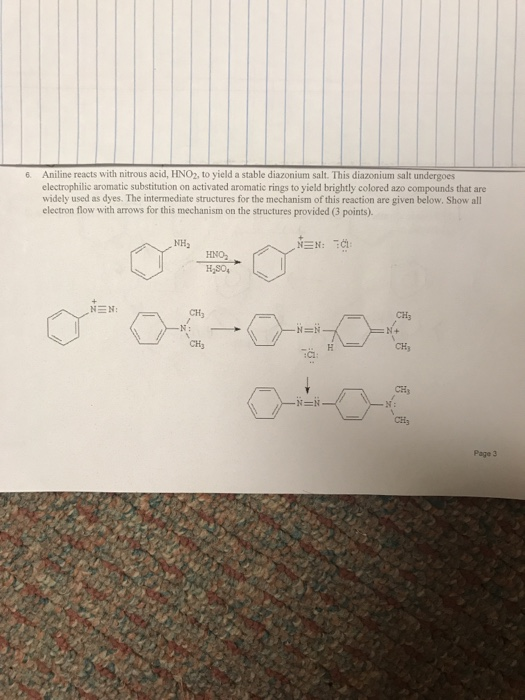
![Solved] Aniline Reacts with Nitrous Acid,HNO2,to Yield a Stable Diazonium Salt | Quiz+ Solved] Aniline Reacts with Nitrous Acid,HNO2,to Yield a Stable Diazonium Salt | Quiz+](https://d2lvgg3v3hfg70.cloudfront.net/TB4944/11eab917_1506_4469_99e6_1f99a6c80b57_TB4944_00.jpg)


![Aniline reacts with conc. \\[\\;{\\text{HN}}{{\\text{O}}_{\\text{3}}}\\] to give:-\n \n \n \n \n Aniline reacts with conc. \\[\\;{\\text{HN}}{{\\text{O}}_{\\text{3}}}\\] to give:-\n \n \n \n \n](https://www.vedantu.com/question-sets/f102ae28-5724-46a5-be68-375d93b118677418549098762929594.png)