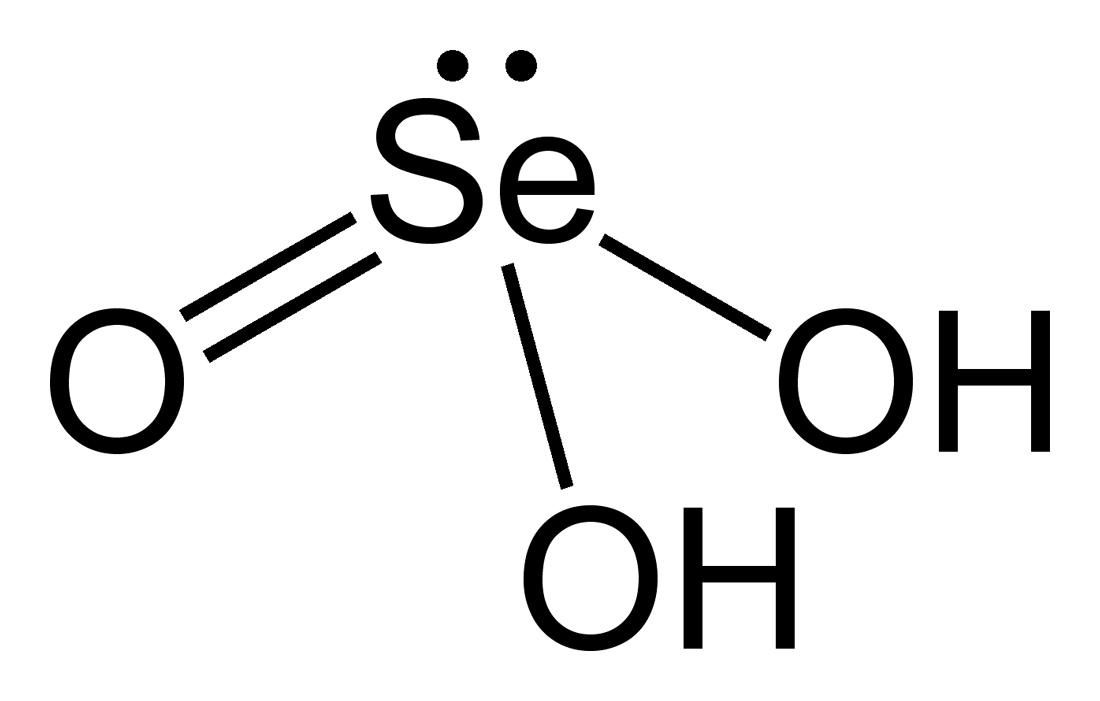
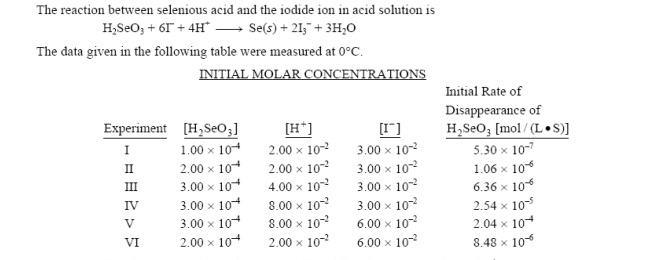
Selenious acid `(H_(2)SeO_(3))`,a diprotoc acid has `Ka_(1)=10^(-6)` and `Ka_(2)=10^(-8)` respectively. Appoximate pH of 0.01M `NaHSeO_(3)` is given by

Selenious acid‐doped polyaniline synthesis and characterization by chemical oxidative solid‐state polymerization of aniline with SeO2 as an oxidizing agent - Perumal - 2022 - Polymer International - Wiley Online Library

6. Illustration of the transformation of selenious acid to selenium... | Download Scientific Diagram