Krisztina Galgoczy - Supply Chain Specialist - TAPI - Teva Active Pharmaceutical Ingredients | LinkedIn

Population Pharmacokinetic Modeling and Simulation of TV‐46000: A Long‐Acting Injectable Formulation of Risperidone - Perlstein - 2022 - Clinical Pharmacology in Drug Development - Wiley Online Library

Teva Pharmaceuticals on LinkedIn: #patientsafetyfirst #safeuseofmedicines #improvingpatientslives #tevapharm

Teva Pharmaceuticals hiring Customer Service Analyst II - 2482 in Debrecen, Hajdú-Bihar, Hungary | LinkedIn

PDF) Unmasking Integration Challenges: The Case of Biogal's Acquisition by Teva Pharmaceutical Industries

Long-term cardiovascular safety of febuxostat compared with allopurinol in patients with gout (FAST): a multicentre, prospective, randomised, open-label, non-inferiority trial - The Lancet

Efficacy and safety of oral panobinostat plus subcutaneous bortezomib and oral dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma (PANORAMA 3): an open-label, randomised, phase 2 study - The

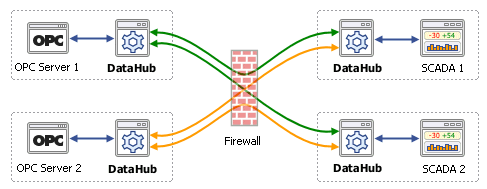
Ops success story teva pharmaceutical hungary final by Konica Minolta Business Solutions Spain, S.A. - Issuu