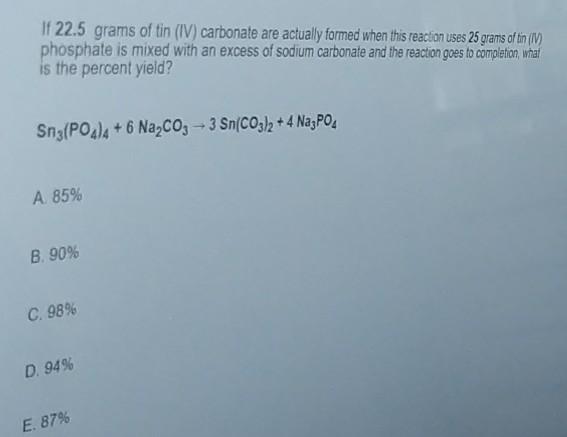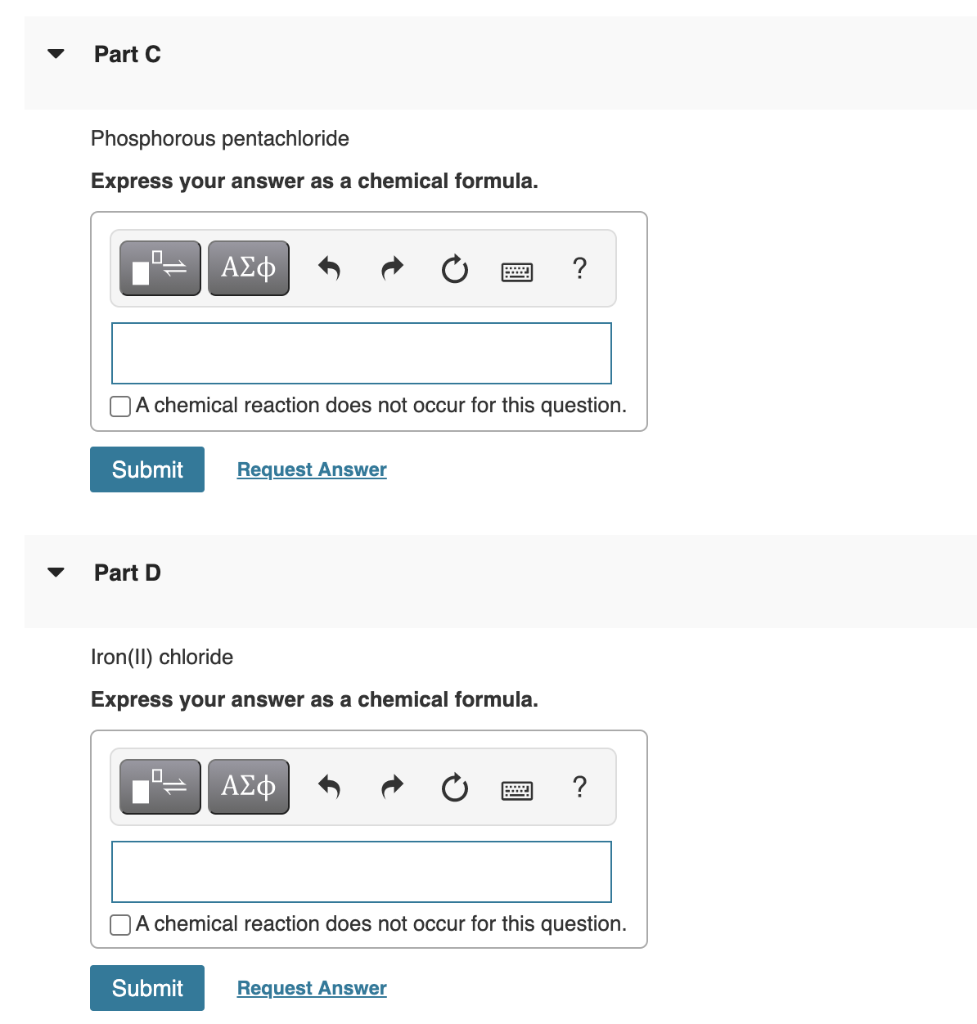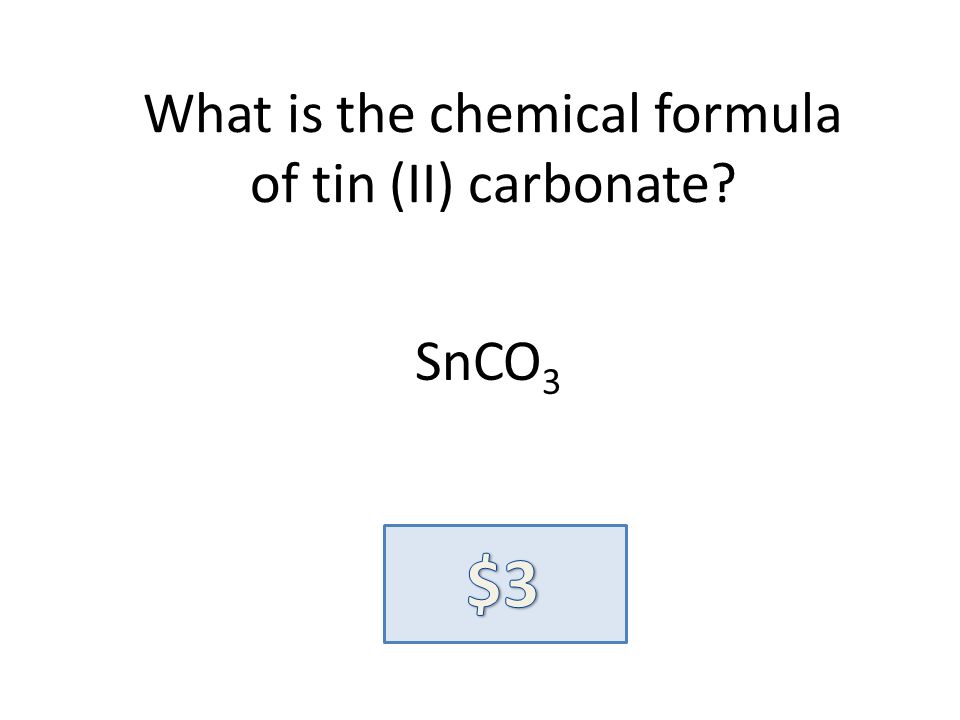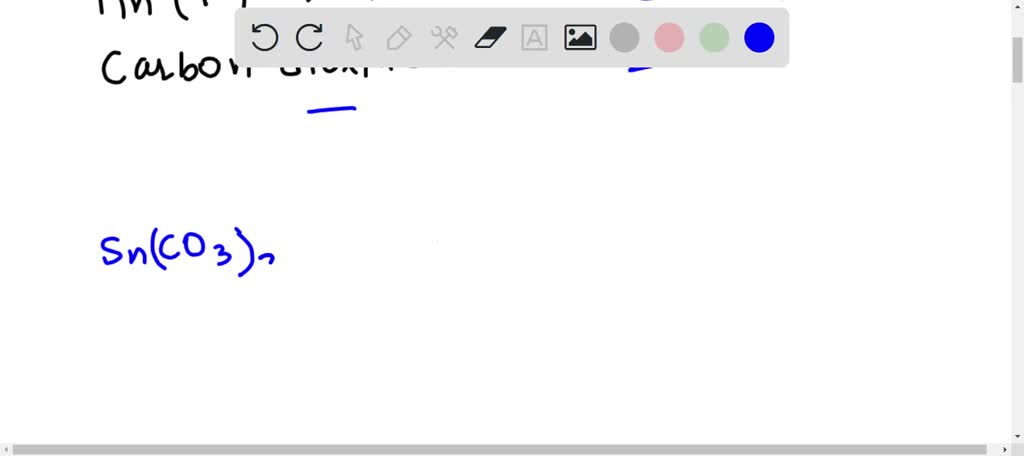
SOLVED: Tin(IV) carbonate decomposes with heat to give solid tin(IV) oxide, and carbon dioxide gas. Express your answer as a chemical equation. Identify all of the phases in your answer.

Mockup Carbonate Containers with a Drink and Condensate on a Background with Shadows Stock Illustration - Illustration of advertising, cylinder: 187499504